
If you are in the market for a softener system, you are bound to have run across the questions revolving around softeners, and wondered “Are softeners bad for the environment?”
This is a very interesting question that I would have to answer with a resounding, “It depends!”
There’s really a lot of things to consider and before an individual can truly pass judgment on a softener’s eco-friendliness,
one must first understand the known potential hazards softeners can have on the environment, their inner workings, the benefits and all the unique considerations for many of these areas.
This in-depth article seeks to provide clarity on this important issue to help you answer the question, are softeners bad for the environment?
The Environmental Implications of Water Softeners
Water softeners, despite their widespread use by homeowners around the world, are not without controversy.
As more and more products hit the market and more households are looking to address hard water issues they may be experiencing, the conversation has shifted from the benefits they provide to their environmental footprint and the repercussions of their use.
What are some of the known negative impacts softeners have on the environment?
Salt Discharge
The primary agent responsible for the softening of water in most traditional softeners is sodium chloride better known as salt.
The softening process results in brine, a concentrated salt byproduct, which finds its way into our waterways, as well as land.
Elevated salt concentrations in freshwater sources can wreak havoc on the local ecosystem. From affecting freshwater fish species to altering the natural habitat of certain aquatic plants, salt discharge is a major environmental concern.
On land, salt can cause just as many issues from preventing plant growth, to harming crops, and potential seepage into groundwater. Over time, saltwater contamination can make the water in these sources unsuitable for both consumption and irrigation. Not good!
Additionally, high amounts of salt water returning to municipalities can cause negative impacts to the processes used to remove unwanted contaminants.
Regeneration
The main reason salt discharge is an issue is because of the regeneration, an essential aspect of the softening process.
As hard minerals accumulate within the softener system, a regeneration process is initiated to flush them out.
While this ensures the continued efficiency of the softener, it also has two primary environmental concerns: the large amounts of water used and the consequent release of high salt concentrations.
Frequent regenerations can significantly increase a household’s water consumption. This is concerning, especially in areas struggling with water scarcity. Moreover, the increased frequency of salt discharge can escalate the environmental harm previously discussed.
Water Consumption
Water softeners, especially older models or systems that have been programmed incorrectly, are not always efficient in their water usage.
During the regeneration process, significant amounts of water are used to flush out the accumulated minerals.
This elevated consumption poses problems in areas where water resources are already stretched thin.
In drought-prone regions, excessive water use can put additional strain on already limited resources. Over time, this can lead to more stringent water restrictions, impacting agriculture, industry, and domestic consumption.
How and Ion Exchange Softener Works
One of the main reasons water softeners are implicated as being potentially harmful to the environment has to do with the way they actually soften or remove calcium and magnesium from water.
Water softeners’ are able to remove calcium and magnesium by way of a process called ion exchange. Even if you aren’t a fan of science, you have to admit the ion exchange process is pretty fascinating.
The main component of a softener system that carries out this process is the resin bed that lies at the bottom of the system’s vessel. A resin bed is composed of hundreds of thousands tiny ionically charged beads known as resin.
When water containing calcium and magnesium also known as hard water flows over the resin beads, the ions of calcium and magnesium are attracted to the resin, and in turn displaces sodium ions residing on the resin.
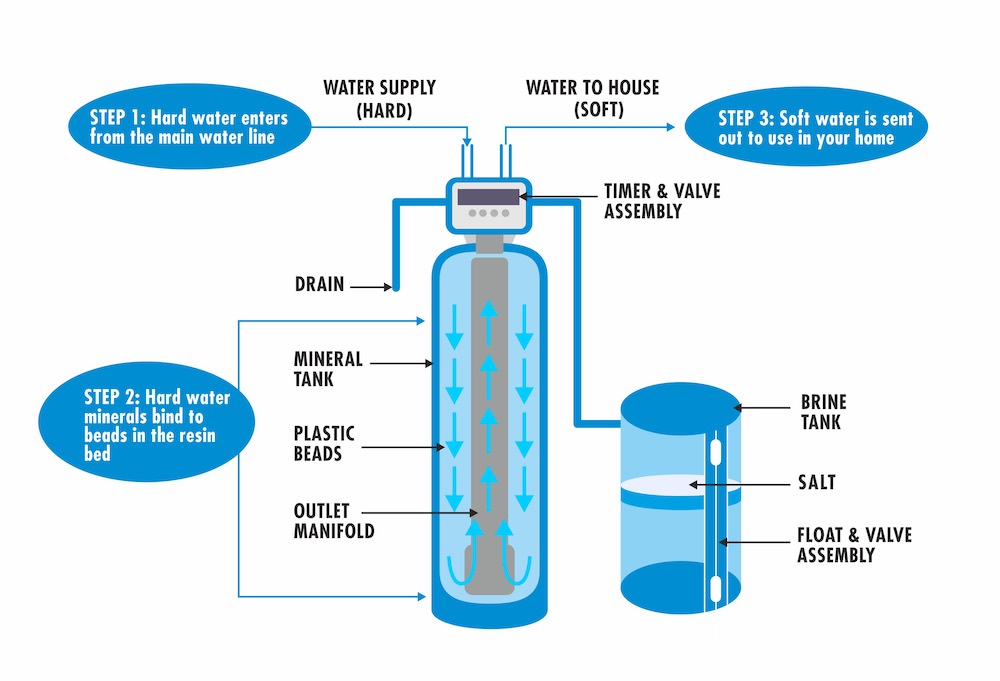
This ion exchange process produces the calcium and magnesium free “soft” water you know and love.
You may be asking yourself, what about those that are getting away from using sodium chloride salt and switching to potassium chloride salt?
Wouldn’t this fix the concerns? In short, no, it isn’t just sodium that’s toxic to both aquatic life and plant life, it’s also chloride. Many of the same concerns with sodium are also concerns with chloride.
Keep Reading: How Does a Water Softener Work?
Eco-Friendly Alternatives To Water Softeners
Recognizing the environmental challenges posed by traditional water softeners, there’s been a big push by municipalities around the country to encourage the use of more sustainable solutions.
Salt-Free Water Softeners
These systems, instead of removing hard minerals, alter their structure to prevent them from depositing as scale. They achieve this without using salt, eliminating the issue of brine discharge. Due to the fact that they don’t use salt in the first place, they also don’t require regeneration which helps save significant amounts of water over the lifetime of the system.

It’s worth noting that beyond their direct environmental benefits, salt-free softeners often require less maintenance.
Regeneration Frequency and Dialing in Regeneration
Over the years there’s been significant advancements in technology that has led to the development of softeners that can intelligently determine the optimal time for regeneration.
They do this by utilizing both internal and/or external water meters that count down the remaining softening capacity of a water softener instead of softeners that regenerate simply based on time which provides pretty significant reductions in unnecessary water and salt use. It also ensures you have high quality water all the time!
Another area of technology improvement with water softeners is the resin. Nowadays, there are many options when it comes to what type and quality of resin you add to your softener system.
These advancements allow for greater resin capacity and its ability to withstand things like chlorine and other contaminants like iron that would otherwise reduce the softener’s efficiency over time and inevitably lead to more frequent regenerations being necessary.
So, before you go throwing out your existing softener system, these may be some viable options for you to help reduce the negative environmental impact your softener has on the environment.
Further Alternative Water Treatment Solutions
While softeners’ negative impacts have been covered extensively, we can’t say all that bad stuff about them and not at least mention the negative impacts other water treatment solutions cause.
Reverse Osmosis
The reverse osmosis process, while incredibly effective in removing a vast array of contaminants, is by no means water efficient. A significant portion of the input water is discarded as waste. In addition to its direct water wastage, the discarded water often carries with it highly concentrated contaminants, which can be an environmental concern if not properly managed.
When using reverse osmosis systems, we recommend using these systems only on the water you are consuming.
Activated Carbon Filters:
These types of filters are great for removing organic compounds, taste, and odor from water. The primary environmental concern here is the disposal of used carbon and the energy-intensive activation process. Over-reliance on activated carbon filters can contribute to increased waste generation, particularly if the used carbon is not properly recycled or repurposed.

Additionally, larger carbon filters that require the media to be loaded into vessels pose a health risk due to the carbon dust created in the loading process. For obvious reasons, inhaling straight carbon dust into the lungs could cause some issues.
Water Distillers
While highly effective in purifying water, distillers are energy-intensive. They work by boiling water and collecting the pure condensate. The energy source for the distillation process determines its environmental impact. Non-renewable energy sources can make this method less eco-friendly.
KDF Media
Utilizing copper and zinc, KDF is effective in removing chlorine and some heavy metals. However, the mining and processing of the metals have environmental implications.Beyond the direct environmental costs of metal extraction, there’s also the broader issue of the finite nature of these resources.
Cation Exchange Resin
While similar in function to ion-exchange resins used in softeners, cation exchange resins find application in more specialized water purification processes. Their environmental considerations center around the concentrated acid and caustic chemicals used in the regeneration process. The release of these chemicals can pose environmental risks, especially if they find their way into natural water sources.
Final Thoughts
Water softeners undeniably offer households significant benefits by addressing hard water problems. However, like many conveniences in the modern world, they come with environmental trade-offs. The salt discharge and increased water consumption, particularly during the regeneration process, have tangible environmental impacts, especially on freshwater ecosystems and in areas experiencing water scarcity.
That being said, the continued advancements in technology and increased environmental awareness have spurred the development of more efficient and eco-friendly alternatives. Measures such as optimizing regeneration cycles and adopting salt-free water softeners can mitigate some of the environmental concerns.
While traditional water softeners do have negative environmental impacts, informed choices, responsible use, and technological advancements can reduce their ecological footprint. It’s essential for consumers to evaluate what they truly need and be aware of these impacts and consider eco-friendlier alternatives and practices where possible.


